Page 10 - Demo-1
P. 10
Br
Br— Br—
Ph
O Br
O O
OH
Br
O cat OH cat Br O
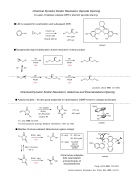
Chemical Dynamic Kinetic Resolution: Epoxide Opening Co-salen complexes catalyze DKR in phenolic epoxide opening
! LiBr is required for racemization and subsequent DKR
OH
OHBr Ph ButOOtBu
O 4 mol% cat. O
Br Co tBu
PhOH
fast
PhOH
slow
Ph
Ph O
Ph
PhOH
slow
Ph PhOH
fast
O Ph
Jacobsen, JACS, 1999, 121, 6086.
4 mol% LiBr CH3CN, MS 3A
74% yield > 99% ee
But
! Exceptionally high ee attributed to further resolution of minor product
O cat OH cat OH
Chemical Dynamic Kinetic Resolution: Azlactone and Dioxolanedione Opening ! Azlactones (pKa ~ 9) have good propensity for racemization; DMAP known to catalyze alcoholysis
R
O
R
O
O
EtOH NR3* R
fast OH
OEt
OEt
R=aryl: 91-95%ee 68 - 85% yield
N H
Et
OO HH
N
O
Chiral amine catalyzes both racemization and alcoholysis of dioxolanediones
catalyst
N
Ph toluene, RT
O
O
NR3* (10 mol%)
—78 °C
R = alkyl:
No DKR (due to decreased acidity)
MeO
O O
EtOH NR3* R
slow
OH
5% catalyst
iPrOH
10% PhCOOH
OiPr 61% ee Ph 94% yield
HN
Fu, JOC, 1998, 63, 3154.
For other azlactone openings: Seebach, Tetrahedron, 1997, 53, 7539.
! Modifies Cinchona alkaloid: Bifunctional organic catalyst OO
Fe Me Me Me
Me
catalyst
O
O Me2N
O
ON
Deng, JACS, 2002, 124, 2870. Lactone opening: Bringmann, Acc. Chem. Res., 2001, 34, 615.
NN
catalyst