Page 258 - Fluid, Electrolyte, and Acid-Base Disorders in Small Animal Practice
P. 258
Introduction to Acid-Base Disorders 249
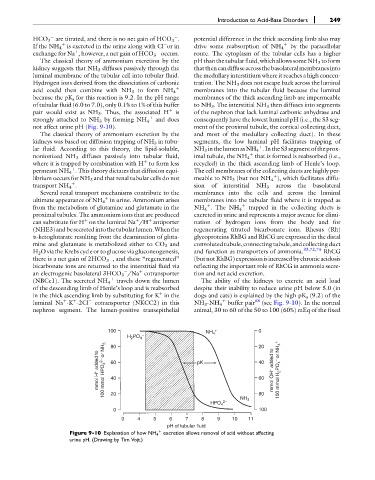
HCO 3 are titrated, and there is no net gain of HCO 3 . potential difference in the thick ascending limb also may
þ
If the NH 4 is excreted in the urine along with Cl or in drive some reabsorption of NH 4 þ by the paracellular
exchange for Na , however, a net gain of HCO 3 occurs. route. The cytoplasm of the tubular cells has a higher
þ
The classical theory of ammonium excretion by the pHthan thetubular fluid, which allowssomeNH 3 toform
kidney suggests that NH 3 diffuses passively through the thatthencandiffuseacrossthebasolateralmembranesinto
luminal membrane of the tubular cell into tubular fluid. the medullary interstitium where it reaches a high concen-
Hydrogen ions derived from the dissociation of carbonic tration. The NH 3 does not escape back across the luminal
þ
acid could then combine with NH 3 to form NH 4 membranes into the tubular fluid because the luminal
because the pK a for this reaction is 9.2. In the pH range membranes of the thick ascending limb are impermeable
of tubular fluid (6.0 to 7.0), only 0.1% to 1% of this buffer to NH 3 . The interstitial NH 3 then diffuses into segments
pair would exist as NH 3 . Thus, the associated H þ is of the nephron that lack luminal carbonic anhydrase and
strongly attached to NH 3 by forming NH 4 and does consequently have the lowest luminal pH (i.e., the S3 seg-
þ
not affect urine pH (Fig. 9-10). ment of the proximal tubule, the cortical collecting duct,
The classical theory of ammonium excretion by the and most of the medullary collecting duct). In these
kidneys was based on diffusion trapping of NH 3 in tubu- segments, the low luminal pH facilitates trapping of
þ
lar fluid. According to this theory, the lipid-soluble, NH 3 inthelumenasNH 4 .IntheS3segmentoftheprox-
þ
nonionized NH 3 diffuses passively into tubular fluid, imal tubule, the NH 4 that is formed is reabsorbed (i.e.,
þ
where it is trapped by combination with H to form less recycled) in the thick ascending limb of Henle’s loop.
þ
permeant NH 4 . This theory dictates that diffusion equi- The cell membranes of the collecting ducts are highly per-
þ
librium occurs for NH 3 and that renal tubular cells do not meable to NH 3 (but not NH 4 ), which facilitates diffu-
þ
transport NH 4 . sion of interstitial NH 3 across the basolateral
Several renal transport mechanisms contribute to the membranes into the cells and across the luminal
ultimate appearance of NH 4 in urine. Ammonium arises membranes into the tubular fluid where it is trapped as
þ
from the metabolism of glutamine and glutamate in the NH 4 . The NH 4 þ trapped in the collecting ducts is
þ
proximal tubules. The ammonium ions that are produced excreted in urine and represents a major avenue for elimi-
þ
can substitute for H on the luminal Na /H antiporter nation of hydrogen ions from the body and for
þ
þ
(NHE3)andbesecretedintothetubularlumen.Whenthe regenerating titrated bicarbonate ions. Rhesus (Rh)
a-ketoglutarate resulting from the deamination of gluta- glycoproteins RhBG and RhCG are expressed in the distal
mine and glutamate is metabolized either to CO 2 and convolutedtubule,connectingtubule,andcollectingduct
H 2 O via the Krebs cycle or toglucose via gluconeogenesis, and function as transporters of ammonia. 33,72,74 RhCG
there is a net gain of 2HCO 3 , and these “regenerated” (but not RhBG) expression is increasedby chronic acidosis
bicarbonate ions are returned to the interstitial fluid via reflecting the important role of RhCG in ammonia secre-
an electrogenic basolateral 3HCO 3 /Na cotransporter tion and net acid excretion.
þ
(NBCe1). The secreted NH 4 travels down the lumen The ability of the kidneys to excrete an acid load
þ
of the descending limb of Henle’s loop and is reabsorbed despite their inability to reduce urine pH below 5.0 (in
in the thick ascending limb by substituting for K in the dogs and cats) is explained by the high pK a (9.2) of the
þ
þ
þ
þ
luminal Na -K -2Cl cotransporter (NKCC2) in this NH 3 -NH 4 buffer pair 68 (see Fig. 9-10). In the normal
nephron segment. The lumen-positive transepithelial animal, 30 to 60 of the 50 to 100 (60%) mEq of the fixed
100 NH + 0
H PO 4 – 4 20
2
80
mmol H + added to 100 mmol HPO 4 2– or NH 3 60 pK 40 mmol OH – added to 100 mmol H 2 PO 4 – or NH 4 +
60
40
20
HPO 4 2– NH 3 80
0 100
0 4 5 6 7 8 9 10 11
pH of tubular fluid
Figure 9-10 Explanation of how NH 4 excretion allows removal of acid without affecting
þ
urine pH. (Drawing by Tim Vojt.)