Page 42 - The Toxicology of Fishes
P. 42
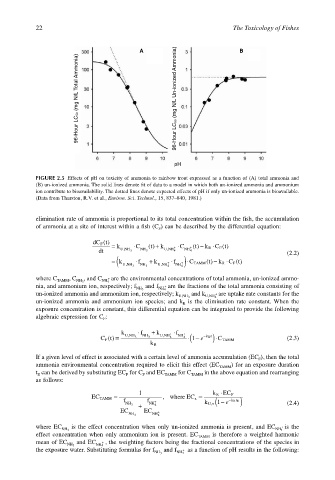
22 The Toxicology of Fishes
A B
96-Hour LC50 (mg N/L Total Ammonia) 96-Hour LC50 (mg N/L Un-ionized Ammonia)
pH
FIGURE 2.5 Effects of pH on toxicity of ammonia to rainbow trout expressed as a function of (A) total ammonia and
(B) un-ionized ammonia. The solid lines denote fit of data to a model in which both un-ionized ammonia and ammonium
ion contribute to bioavailability. The dotted lines denote expected effects of pH if only un-ionized ammonia is bioavailable.
(Data from Thurston, R.V. et al., Environ. Sci. Technol., 15, 837–840, 1981.)
elimination rate of ammonia is proportional to its total concentration within the fish, the accumulation
of ammonia at a site of interest within a fish (C ) can be described by the differential equation:
F
dC t() = k ⋅ C t () + k ⋅ C t () − k R ⋅⋅Ct()
F
,
,
dt UNH 3 NH 3 U NH 4 + NH 4 + F (2.2)
(
= k ⋅f + k + ⋅f + ) ⋅C TAMM ( − k R ⋅C F ( )
)
t
t)
,
,
UNH 3 NH 3 U NH 4 NH 4
where C TAMM , C NH 3 , and C NH 4 + are the environmental concentrations of total ammonia, un-ionized ammo-
nia, and ammonium ion, respectively; f and f + are the fractions of the total ammonia consisting of
NH 3 NH 4
un-ionized ammonia and ammonium ion, respectively; k and k + are uptake rate constants for the
U,NH 3 U,NH 4
un-ionized ammonia and ammonium ion species; and k is the elimination rate constant. When the
R
exposure concentration is constant, this differential equation can be integrated to provide the following
algebraic expression for C :
F
k f ⋅ + k + f ⋅ +
Ct() = UNH 3 NH 3 U NH 4 NH 4 ⋅(1 e − kt ) ⋅⋅C TAMM (2.3)
−
,
,
R
F
k R
If a given level of effect is associated with a certain level of ammonia accumulation (EC ), then the total
F
ammonia environmental concentration required to elicit this effect (EC TAMM ) for an exposure duration
t can be derived by substituting EC for C and EC TAMM for C TAMM in the above equation and rearranging
F
E
F
as follows:
C
EC TAMM = 1 , where EC x = k R ⋅ EC F
f f + k Ux , 1− ( e − kt )
RE
NH 3 + NH 4 (2.4)
EC EC +
NH 3 NH 4
where EC is the effect concentration when only un-ionized ammonia is present, and EC + is the
NH 3 NH 4
effect concentration when only ammonium ion is present. EC TAMM is therefore a weighted harmonic
mean of EC and EC + , the weighting factors being the fractional concentrations of the species in
NH 3 NH4
the exposure water. Substituting formulas for f and f + as a function of pH results in the following:
NH 3 NH 4