Page 61 - The Toxicology of Fishes
P. 61
Bioavailability of Chemical Contaminants in Aquatic Systems 41
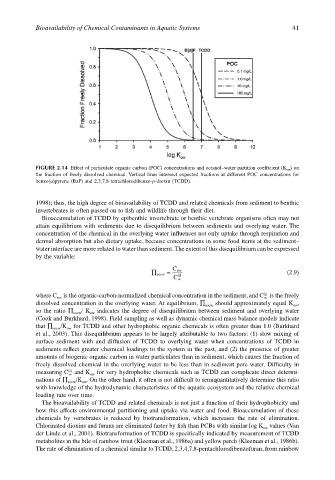
FIGURE 2.14 Effect of particulate organic carbon (POC) concentrations and octanol–water partition coefficient (K ow ) on
the fraction of freely dissolved chemical. Vertical lines intersect expected fractions at different POC concentrations for
benzo(a)pyrene (BaP) and 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD).
1998); thus, the high degree of bioavailability of TCDD and related chemicals from sediment to benthic
invertebrates is often passed on to fish and wildlife through their diet.
Bioaccumulation of TCDD by epibenthic invertebrate or benthic vertebrate organisms often may not
attain equilibrium with sediments due to disequilibrium between sediments and overlying water. The
concentration of the chemical in the overlying water influences not only uptake through respiration and
dermal absorption but also dietary uptake, because concentrations in some food items at the sediment–
water interface are more related to water than sediment. The extent of this disequilibrium can be expressed
by the variable:
= C soc (2.9)
∏ socw fd
C w
fd
where C is the organic-carbon-normalized chemical concentration in the sediment, and C is the freely
soc
w
dissolved concentration in the overlying water. At equilibrium, ∏ socw should approximately equal K ,
ow
so the ratio ∏ socw / K indicates the degree of disequilibrium between sediment and overlying water
ow
(Cook and Burkhard, 1998). Field sampling as well as dynamic chemical mass balance models indicate
that ∏ socw /K for TCDD and other hydrophobic organic chemicals is often greater than 1.0 (Burkhard
ow
et al., 2003). This disequilibrium appears to be largely attributable to two factors: (1) slow mixing of
surface sediment with and diffusion of TCDD to overlying water when concentrations of TCDD in
sediments reflect greater chemical loadings to the system in the past, and (2) the presence of greater
amounts of biogenic organic carbon in water particulates than in sediment, which causes the fraction of
freely dissolved chemical in the overlying water to be less than in sediment pore water. Difficulty in
fd
measuring C and K for very hydrophobic chemicals such as TCDD can complicate direct determi-
ow
w
nations of ∏ /K . On the other hand, it often is not difficult to semiquantitatively determine this ratio
socw ow
with knowledge of the hydrodynamic characteristics of the aquatic ecosystem and the relative chemical
loading rate over time.
The bioavailability of TCDD and related chemicals is not just a function of their hydrophobicity and
how this affects environmental partitioning and uptake via water and food. Bioaccumulation of these
chemicals by vertebrates is reduced by biotransformation, which increases the rate of elimination.
Chlorinated dioxins and furans are eliminated faster by fish than PCBs with similar log K values (Van
ow
der Linde et al., 2001). Biotransformation of TCDD is specifically indicated by measurement of TCDD
metabolites in the bile of rainbow trout (Kleeman et al., 1986a) and yellow perch (Kleeman et al., 1986b).
The rate of elimination of a chemical similar to TCDD, 2,3,4,7,8-pentachlorodibenzofuran, from rainbow