Page 54 - The Toxicology of Fishes
P. 54
34 The Toxicology of Fishes
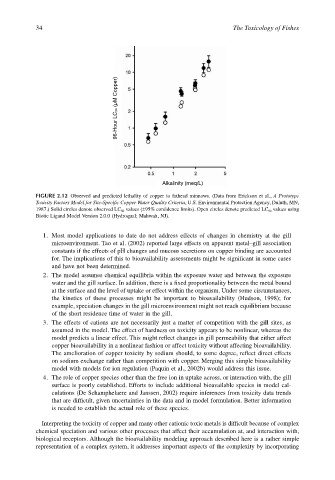
96-Hour LC50 (µM Copper)
Alkalinity (meq/L)
FIGURE 2.12 Observed and predicted lethality of copper to fathead minnows. (Data from Erickson et al., A Prototype
Toxicity Factors Model for Site-Specific Copper Water Quality Criteria, U.S. Environmental Protection Agency, Duluth, MN,
1987.) Solid circles denote observed LC 50 values (±95% confidence limits). Open circles denote predicted LC 50 values using
Biotic Ligand Model Version 2.0.0 (Hydroqual; Mahwah, NJ).
1. Most model applications to date do not address effects of changes in chemistry at the gill
microenvironment. Tao et al. (2002) reported large effects on apparent metal–gill association
constants if the effects of pH changes and mucous secretions on copper binding are accounted
for. The implications of this to bioavailability assessments might be significant in some cases
and have not been determined.
2. The model assumes chemical equilibria within the exposure water and between the exposure
water and the gill surface. In addition, there is a fixed proportionality between the metal bound
at the surface and the level of uptake or effect within the organism. Under some circumstances,
the kinetics of these processes might be important to bioavailability (Hudson, 1998); for
example, speciation changes in the gill microenvironment might not reach equilibrium because
of the short residence time of water in the gill.
3. The effects of cations are not necessarily just a matter of competition with the gill sites, as
assumed in the model. The effect of hardness on toxicity appears to be nonlinear, whereas the
model predicts a linear effect. This might reflect changes in gill permeability that either affect
copper bioavailability in a nonlinear fashion or affect toxicity without affecting bioavailability.
The amelioration of copper toxicity by sodium should, to some degree, reflect direct effects
on sodium exchange rather than competition with copper. Merging this simple bioavailability
model with models for ion regulation (Paquin et al., 2002b) would address this issue.
4. The role of copper species other than the free ion in uptake across, or interaction with, the gill
surface is poorly established. Efforts to include additional bioavailable species in model cal-
culations (De Schamphelaere and Janssen, 2002) require inferences from toxicity data trends
that are difficult, given uncertainties in the data and in model formulation. Better information
is needed to establish the actual role of these species.
Interpreting the toxicity of copper and many other cationic toxic metals is difficult because of complex
chemical speciation and various other processes that affect their accumulation at, and interaction with,
biological receptors. Although the bioavailability modeling approach described here is a rather simple
representation of a complex system, it addresses important aspects of the complexity by incorporating