Page 13 - PD-L1 EbookV2 Flip PDF
P. 13
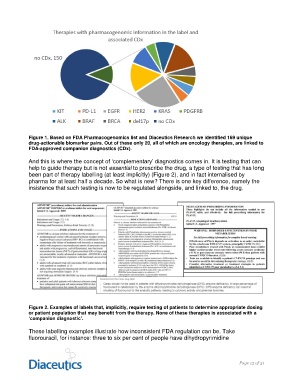
Figure 1. Based on FDA Pharmacogenomics list and Diaceutics Research we identified 169 unique
drug-actionable biomarker pairs. Out of these only 20, all of which are oncology therapies, are linked to
FDA-approved companion diagnostics (CDx).
And this is where the concept of ‘complementary’ diagnostics comes in. It is testing that can
help to guide therapy but is not essential to prescribe the drug, a type of testing that has long
been part of therapy labelling (at least implicitly) (Figure 2), and in fact internalised by
pharma for at least half a decade. So what is new? There is one key difference, namely the
insistence that such testing is now to be regulated alongside, and linked to, the drug.
Figure 2. Examples of labels that, implicitly, require testing of patients to determine appropriate dosing
or patient population that may benefit from the therapy. None of these therapies is associated with a
‘companion diagnostic’.
These labelling examples illustrate how inconsistent FDA regulation can be. Take
fluorouracil, for instance: three to six per cent of people have dihydropyrimidine
Page 13 of 31