Page 30 - Demo-1
P. 30
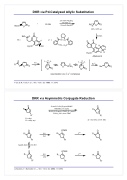
DKR via Pd-Catalyzed Allylic Substitution
+
R OH
2.5 mol% Pd2dba3
7.5mol%L* RO
O
89%, >95% ee
BocO O
EtO O
30 mol% Bu4NCl
H O
O
O
OOO
R= IL*=NHHN OO
6 steps
O H
O OH
H3CO
OH
PPh2 Ph2P
O
Pd
L Pd L
L Pd L
H3CO
Aflatoxin B
RO OL* O
Nu
O OPdL2
OO OO
racemization via η3-η1 complexes
Nu O O
Trost, B. M.; Toste, F. D. J. Am. Chem. Soc. 1999, 121, 3543
DKR via Asymmetric Conjugate Reduction
O R
R'
10 mol% CuCl/(S)-p-tol-BINAP NaOt-Bu (1.7 equiv)
tBuOH (5.0 equiv), PMHS (2.2 equiv) PhCH3, 26 h, then TBAF
O R
R'
R = alkyl
R' = alkyl, aryl
de = 82-93%, ee=91-94%
O OTMS O R [H] R F R
fast
R' R' R' NaOR, ROH very fast
O
R [H] R F
slow
R' R'
OTMS
O R
R'
Jurkauskas, V.; Buchwald, S. L. J. Am. Chem. Soc. 2002, 124, 2892