Page 27 - Demo-1
P. 27
Me N
Me Ph
Me N N Me Ph
R
enantioenriched
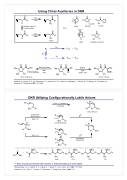
Using Chiral Auxiliaries in DKR
O Nu O O O O
H O H3CN N O ON
CO2tBu Me Durst Nunami Devine
R* = OOO
R* Nu R* H3CN N Fast SN
X Nu O2 MePh
R* slow R* X Nu
addition-induced epimerization
major
S kR P+P
R RRRS
kinv
S kS P+P
OOOOOO
N Me NaCH(CO2Me)2
Me
BnNH2
TBAI Et3N/THF
Me N N Me
NHBn Me Ph
100%, 74% de
CH(CO Me) TBAI 2 2 THF
78%, 40-60% de
Br
Ph
Boc
N
n-BuLi / (-)-sparteine PhCH2Br
H Ph Ph Ar
N
S SRSS
Caddick, S.; Afonso, C. A. M.; Candeias, S. X.; Hitchcock, P. B.; Jenkins, K.; Murtagh, L.; Pardoe, D.; Gil Santos, A.; Treweeke, N.; Weaving, R. Tetrahedron 2001, 57, 6589
DKR Utilizing Configurationally Labile Anions
Ar
Boc
asymmetric deprotonation
RLi / L*
R
L* Li Boc
E
84%, 96% ee
R
EAr N Boc
H HH
Ar N Boc R
L*Li N
RLi / L* Boc R
L*Li N Boc
Ar
Ar
E
asymmetric substitution
N
Ar
NBn2 NBn2
+H Ph Ph PhPh PhPh PhPh Ph
NBn2
PhMe O OH OH OH OH
Li
Ratio of products identical with racemic or enatiomerically pure electrophile
Weisenburger, G. A.; Faibish, N. C.; Pippel, D. J.; Beak, P. J. Am. Chem. Soc. 1999, 121, 9522 Hoffmann, R. W.; Ruhl, T.; Chemla, F.; Zahneisen, T. Liebigs Ann. Chem. 1992, 719
NBn2 NBn2
Ward Caddick & Jenkins
60% 21% 13% 6%