Page 25 - Demo-1
P. 25
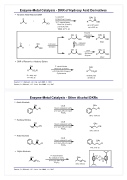
Enzyme-Metal Catalysis - DKR of Hydroxy Acid Derivatives
Tandem Aldol Reaction/DKR
O
1) LDA/THF
2) NH4Cl sat. 2 equiv.
3) P. cepacia lipase Shvo Cat. (6 mol%)
p-Cl-C6H4-OAc TBME, 60 oC, 6d
OAc O
O RH OEt
+
R
OEt
up to 82% yield up to 99% ee
Enzyme
OH O Acyl donor OAc O
R OEt Fast R OEt
O
+
RH OEt
Ru - catalyzed racemization
OH O
Major
OAc O
OEt
Minor
OLi C-C bond
formation
R
OEt
Slow
Acyl donor R Enzyme
DKR of Racemic α-Hydroxy Esters OH
P. cepacia lipase Shvo Cat. (2 mol%)
p-Cl-C6H4-OAc (3 equiv) Cyclohexane
OAc
R CO2R'
60-80% yield 30-98% ee
R
CO2R'
R = alkyl, aryl R' = Me, Et
Huerta, F. F.; Backvall, J.-E. Org. Lett. 2001, 3, 1209 Pamies, O.; Backvall, J.-E. Chem. Rev. 2003, 103, 3247
Enzyme-Metal Catalysis - Other Alcohol DKRs
Azido Alcohols
Hydroxy Nitriles
Halo Alcohols
Allylic Alcohols
OH CALB
OAc
N3
86%, >99% ee
OAc
CN
7
93%, 92% ee
OAc
Cl
74%, 91% ee
N3
OH
CN
Shvo's catalyst (4 mol%)
p-Cl-C6H4-OAc (3 equiv) PhCH3
CALB
Shvo's catalyst (4 mol%)
p-Cl-C6H4-OAc (3 equiv) PhCH3
CALB
Shvo's catalyst (4 mol%)
p-Cl-C6H4-OAc (3 equiv) PhCH3
P. cepacia lipase Ru catalyst (4 mol%)
p-Cl-C6H4-OAc, Et3N CH2Cl2
7
OH
OH
Pr
Cl
OAc
Pr
83%, 95% ee
Cl Cl
Ru H Ru
Cl
Pamies, O.; Backvall, J.-E. Chem. Rev. 2003, 103, 3247