Page 249 - Fluid, Electrolyte, and Acid-Base Disorders in Small Animal Practice
P. 249
240 ACID-BASE DISORDERS
[H ] + 120 100 80 70
pH 6.92 7.00 60
100 7.10 7.15 7.22 34
50
+
7.30
32
40
[H ] nEq/L
Acute resp. acidosis Chronic resp. acidosis 7.52 HCO 3 mEq/L 28
80 7.40 30 30 Whole blood
PaCO 2 mm Hg 40 Metabolic alkalosis 7.70 20 26 Whole body
60
24
Chronic resp. alkalosis
Metabolic acidosis Acute resp. alkalosis 8.00 10 22
20 20
40 60 80 100 120 140
+ PaCO 2
[H ] = 24 pCO 2 (mm Hg)
[HCO 3 ] –
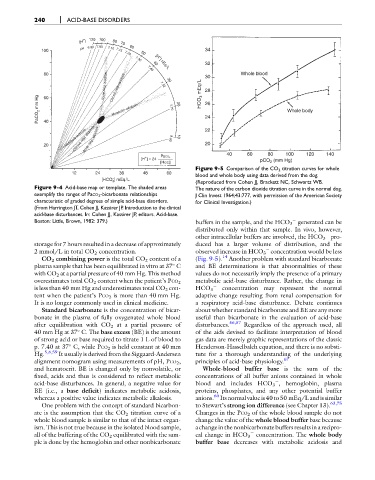
Figure 9-5 Comparison of the CO 2 titration curves for whole
12 24 36 48 60 blood and whole body using data derived from the dog.
–
[HCO ] mEq/L (Reproduced from Cohen JJ, Brackett NC, Schwartz WB.
3
Figure 9-4 Acid-base map or template. The shaded areas The nature of the carbon dioxide titration curve in the normal dog.
exemplify the ranges of PaCO 2 -bicarbonate relationships J Clin Invest 1964;43:777, with permission of the American Society
characteristic of graded degrees of simple acid-base disorders. for Clinical Investigation.)
(From Harrington JT, Cohen JJ, Kassirer JP. Introduction to the clinical
acid-base disturbances. In: Cohen JJ, Kassirer JP, editors. Acid-base.
Boston: Little, Brown, 1982: 379.) buffers in the sample, and the HCO 3 generated can be
distributed only within that sample. In vivo, however,
other intracellular buffers are involved, the HCO 3 pro-
storage for 7 hours resulted in a decrease of approximately duced has a larger volume of distribution, and the
2 mmol/L in total CO 2 concentration. observed increase in HCO 3 concentration would be less
CO 2 combining power is the total CO 2 content of a (Fig. 9-5). 14 Another problem with standard bicarbonate
plasma sample that has been equilibrated in vitro at 37 C and BE determinations is that abnormalities of these
with CO 2 at a partial pressure of 40 mm Hg. This method values do not necessarily imply the presence of a primary
metabolic acid-base disturbance. Rather, the change in
overestimates total CO 2 content when the patient’s PCO 2
is less than 40 mm Hg and underestimates total CO 2 con- HCO 3 concentration may represent the normal
tent when the patient’s PCO 2 is more than 40 mm Hg. adaptive change resulting from renal compensation for
It is no longer commonly used in clinical medicine. a respiratory acid-base disturbance. Debate continues
Standard bicarbonate is the concentration of bicar- about whether standard bicarbonate and BE are any more
bonate in the plasma of fully oxygenated whole blood useful than bicarbonate in the evaluation of acid-base
after equilibration with CO 2 at a partial pressure of disturbances. 56,57 Regardless of the approach used, all
40 mm Hg at 37 C. The base excess (BE) is the amount of the aids devised to facilitate interpretation of blood
of strong acid or base required to titrate 1 L of blood to gas data are merely graphic representations of the classic
p. 7.40 at 37 C, while PCO 2 is held constant at 40 mm Henderson-Hasselbalch equation, and there is no substi-
Hg. 5,6,59 It usually is derived from the Siggaard-Andersen tute for a thorough understanding of the underlying
alignment nomogram using measurements of pH, PCO 2 , principles of acid-base physiology. 67
and hematocrit. BE is changed only by nonvolatile, or Whole-blood buffer base is the sum of the
fixed, acids and thus is considered to reflect metabolic concentrations of all buffer anions contained in whole
acid-base disturbances. In general, a negative value for blood and includes HCO 3 , hemoglobin, plasma
BE (i.e., a base deficit) indicates metabolic acidosis, proteins, phosphates, and any other potential buffer
60
whereas a positive value indicates metabolic alkalosis. anions. Itsnormalvalue is40to50 mEq/L andissimilar
One problem with the concept of standard bicarbon- to Stewart’s strong ion difference (see Chapter 13). 63,75
ate is the assumption that the CO 2 titration curve of a Changes in the PCO 2 of the whole blood sample do not
whole blood sample is similar to that of the intact organ- change the value of the whole blood buffer base because
ism. This is not true because in the isolated blood sample, achange in the nonbicarbonate buffers results in a recipro-
all of the buffering of the CO 2 equilibrated with the sam- cal change in HCO 3 concentration. The whole body
ple is done by the hemoglobin and other nonbicarbonate buffer base decreases with metabolic acidosis and