Page 49 - The Toxicology of Fishes
P. 49
Bioavailability of Chemical Contaminants in Aquatic Systems 29
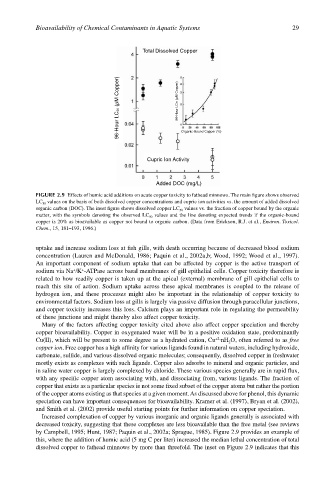
Total Dissolved Copper
96-Hour LC50 (µM Copper) 96-Hour LC50 (µM Copper) Organic-Bound Copper (%)
Cupric Ion Activity
Added DOC (mg/L)
FIGURE 2.9 Effects of humic acid additions on acute copper toxicity to fathead minnows. The main figure shows observed
LC 50 values on the basis of both dissolved copper concentrations and cupric ion activities vs. the amount of added dissolved
organic carbon (DOC). The inset figure shows dissolved copper LC 50 values vs. the fraction of copper bound by the organic
matter, with the symbols denoting the observed LC 50 values and the line denoting expected trends if the organic-bound
copper is 20% as bioavailable as copper not bound to organic carbon. (Data from Erickson, R.J. et al., Environ. Toxicol.
Chem., 15, 181–193, 1996.)
uptake and increase sodium loss at fish gills, with death occurring because of decreased blood sodium
concentration (Lauren and McDonald, 1986; Paquin et al., 2002a,b; Wood, 1992; Wood et al., 1997).
An important component of sodium uptake that can be affected by copper is the active transport of
+
+
sodium via Na /K -ATPase across basal membranes of gill epithelial cells. Copper toxicity therefore is
related to how readily copper is taken up at the apical (external) membrane of gill epithelial cells to
reach this site of action. Sodium uptake across these apical membranes is coupled to the release of
hydrogen ion, and these processes might also be important in the relationship of copper toxicity to
environmental factors. Sodium loss at gills is largely via passive diffusion through paracellular junctions,
and copper toxicity increases this loss. Calcium plays an important role in regulating the permeability
of these junctions and might thereby also affect copper toxicity.
Many of the factors affecting copper toxicity cited above also affect copper speciation and thereby
copper bioavailability. Copper in oxygenated water will be in a positive oxidation state, predominantly
+2
Cu(II), which will be present to some degree as a hydrated cation, Cu ·nH O, often referred to as free
2
copper ion. Free copper has a high affinity for various ligands found in natural waters, including hydroxide,
carbonate, sulfide, and various dissolved organic molecules; consequently, dissolved copper in freshwater
mostly exists as complexes with such ligands. Copper also adsorbs to mineral and organic particles, and
in saline water copper is largely complexed by chloride. These various species generally are in rapid flux,
with any specific copper atom associating with, and dissociating from, various ligands. The fraction of
copper that exists as a particular species is not some fixed subset of the copper atoms but rather the portion
of the copper atoms existing as that species at a given moment. As discussed above for phenol, this dynamic
speciation can have important consequences for bioavailability. Kramer et al. (1997), Bryan et al. (2002),
and Smith et al. (2002) provide useful starting points for further information on copper speciation.
Increased complexation of copper by various inorganic and organic ligands generally is associated with
decreased toxicity, suggesting that these complexes are less bioavailable than the free metal (see reviews
by Campbell, 1995; Hunt, 1987; Paquin et al., 2002a; Sprague, 1985). Figure 2.9 provides an example of
this, where the addition of humic acid (5 mg C per liter) increased the median lethal concentration of total
dissolved copper to fathead minnows by more than threefold. The inset on Figure 2.9 indicates that this