Page 51 - The Toxicology of Fishes
P. 51
Bioavailability of Chemical Contaminants in Aquatic Systems 31
96-Hour LC 50 (µM Copper)
Calcium (meq/L)
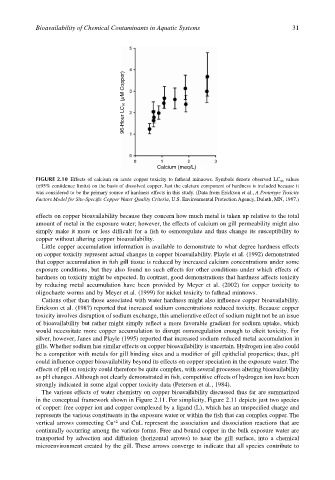
FIGURE 2.10 Effects of calcium on acute copper toxicity to fathead minnows. Symbols denote observed LC 50 values
(±95% confidence limits) on the basis of dissolved copper. Just the calcium component of hardness is included because it
was considered to be the primary source of hardness effects in this study. (Data from Erickson et al., A Prototype Toxicity
Factors Model for Site-Specific Copper Water Quality Criteria, U.S. Environmental Protection Agency, Duluth, MN, 1987.)
effects on copper bioavailability because they concern how much metal is taken up relative to the total
amount of metal in the exposure water; however, the effects of calcium on gill permeability might also
simply make it more or less difficult for a fish to osmoregulate and thus change its susceptibility to
copper without altering copper bioavailability.
Little copper accumulation information is available to demonstrate to what degree hardness effects
on copper toxicity represent actual changes in copper bioavailability. Playle et al. (1992) demonstrated
that copper accumulation in fish gill tissue is reduced by increased calcium concentrations under some
exposure conditions, but they also found no such effects for other conditions under which effects of
hardness on toxicity might be expected. In contrast, good demonstrations that hardness affects toxicity
by reducing metal accumulation have been provided by Meyer et al. (2002) for copper toxicity to
oligochaete worms and by Meyer et al. (1999) for nickel toxicity to fathead minnows.
Cations other than those associated with water hardness might also influence copper bioavailability.
Erickson et al. (1987) reported that increased sodium concentrations reduced toxicity. Because copper
toxicity involves disruption of sodium exchange, this ameliorative effect of sodium might not be an issue
of bioavailability but rather might simply reflect a more favorable gradient for sodium uptake, which
would necessitate more copper accumulation to disrupt osmoregulation enough to elicit toxicity. For
silver, however, Janes and Playle (1995) reported that increased sodium reduced metal accumulation in
gills. Whether sodium has similar effects on copper bioavailability is uncertain. Hydrogen ion also could
be a competitor with metals for gill binding sites and a modifier of gill epithelial properties; thus, pH
could influence copper bioavailability beyond its effects on copper speciation in the exposure water. The
effects of pH on toxicity could therefore be quite complex, with several processes altering bioavailability
as pH changes. Although not clearly demonstrated in fish, competitive effects of hydrogen ion have been
strongly indicated in some algal copper toxicity data (Peterson et al., 1984).
The various effects of water chemistry on copper bioavailability discussed thus far are summarized
in the conceptual framework shown in Figure 2.11. For simplicity, Figure 2.11 depicts just two species
of copper: free copper ion and copper complexed by a ligand (L), which has an unspecified charge and
represents the various constituents in the exposure water or within the fish that can complex copper. The
+2
vertical arrows connecting Cu and CuL represent the association and dissociation reactions that are
continually occurring among the various forms. Free and bound copper in the bulk exposure water are
transported by advection and diffusion (horizontal arrows) to near the gill surface, into a chemical
microenvironment created by the gill. These arrows converge to indicate that all species contribute to