Page 6 - How do I Replenish a Research Participant Payment Fund
P. 6
Submitting A RPPF Replenishment Request
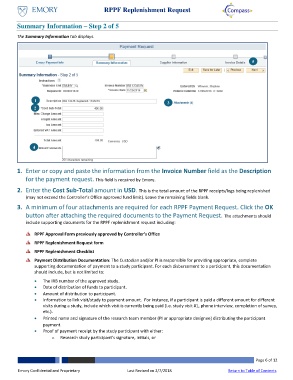
Summary Information – Step 2 of 5
The Summary Information tab displays.
5
1 3
2
4
1. Enter or copy and paste the information from the Invoice Number field as the Description
for the payment request. This field is required by Emory.
2. Enter the Cost Sub-Total amount in USD. This is the total amount of the RPPF receipts/logs being replenished
(may not exceed the Controller’s Office approved fund limit). Leave the remaining fields blank.
3. A minimum of four attachments are required for each RPPF Payment Request. Click the OK
button after attaching the required documents to the Payment Request. The attachments should
include supporting documents for the RPPF replenishment request including:
RPPF Approval Form previously approved by Controller’s Office
RPPF Replenishment Request form
RPPF Replenishment Checklist
Payment Distribution Documentation: The Custodian and/or PI is responsible for providing appropriate, complete
supporting documentation of payment to a study participant. For each disbursement to a participant, this documentation
should include, but is not limited to:
The IRB number of the approved study.
Date of distribution of funds to participant.
Amount of distribution to participant.
Information to link visit/study to payment amount. For instance, if a participant is paid a different amount for different
visits during a study, include which visit is currently being paid (i.e. study visit #1, phone interview, completion of survey,
etc.).
Printed name and signature of the research team member (PI or appropriate designee) distributing the participant
payment.
Proof of payment receipt by the study participant with either:
o Research study participant’s signature, initials, or
Page 6 of 12
Emory Confidential and Proprietary Last Revised on 2/7/2018 Return to Table of Contents