Page 560 - Veterinary Toxicology, Basic and Clinical Principles, 3rd Edition
P. 560
Amitraz Chapter | 41 527
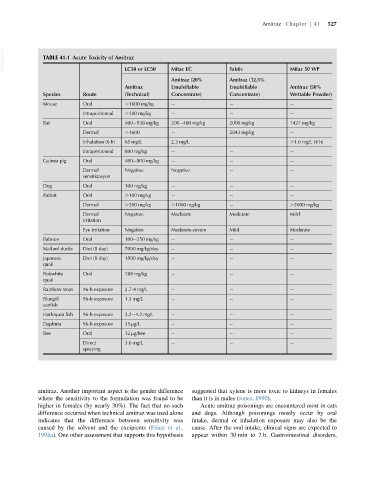
VetBooks.ir TABLE 41.1 Acute Toxicity of Amitraz Mitac EC Taktic Mitac 50 WP
LC50 or LC50
Amitraz (20% Amitraz (12.5%
Amitraz Emulsifiable Emulsifiable Amitraz (50%
Species Route (Technical) Concentrate) Concentrate) Wettable Powder)
Mouse Oral .1600 mg/kg
Intraperitoneal .100 mg/kg
Rat Oral 400 938 mg/kg 200 400 mg/kg 2000 mg/kg 1427 mg/kg
Dermal .1600 2043 mg/kg
Inhalation (6 h) 65 mg/L 2.3 mg/L .1.6 mg/L (4 h)
Intraperitoneal 800 mg/kg
Guinea pig Oral 400 800 mg/kg
Dermal Negative Negative
sensitizasyon
Dog Oral 100 mg/kg
Rabbit Oral .100 mg/kg
Dermal .200 mg/kg .1000 mg/kg .2000 mg/kg
Dermal Negative Moderate Moderate Mild
irritation
Eye irritation Negative Moderate-severe Mild Moderate
Baboon Oral 100 250 mg/kg
Mallard ducks Diet (8 day) 7000 mg/kg/day
Japanese Diet (8 day) 1800 mg/kg/day
quail
Bobwhite Oral 788 mg/kg
quail
Rainbow trout 96-h exposure 2.7-4 mg/L
Bluegill 96-h exposure 1.3 mg/L
sunfish
Harlequin fish 96-h exposure 3.2 4.2 mg/L
Daphnia 96-h exposure 35 μg/L
Bee Oral 12 μg/bee
Direct 3.6 mg/L
spraying
amitraz. Another important aspect is the gender difference suggested that xylene is more toxic to kidneys in females
where the sensitivity to the formulation was found to be than it is in males (Jones, 1990).
higher in females (by nearly 30%). The fact that no such Acute amitraz poisonings are encountered most in cats
difference occurred when technical amitraz was used alone and dogs. Although poisonings mostly occur by oral
indicates that the difference between sensitivity was intake, dermal or inhalation exposure may also be the
caused by the solvent and the excipients (Filazi et al., cause. After the oral intake, clinical signs are expected to
1998a). One other assessment that supports this hypothesis appear within 30 min to 2 h. Gastrointestinal disorders,