Page 319 - Veterinary Immunology, 10th Edition
P. 319
peptide backbone rather than to its amino acid side chains.
VetBooks.ir Nevertheless, structural constraints limit the efficiency of binding of
each allele. As a result, it is likely that only one or two peptides
from an average antigenic protein can bind to any given MHC
molecule. The ability of MHC molecules to bind antigens must be a
limiting factor in generating adaptive immunity and resistance to
infectious agents. Increasing the diversity of MHC molecules
increases the diversity of antigens that can be bound and so
increases resistance to infectious diseases. Because most individuals
are MHC heterozygotes, each individual normally expresses at
most six different class Ia molecules (in humans, for example, two
each are coded for by the HLA-A, -B, and C loci). The number of
expressed MHC molecules is not greater because that would
increase the risk that the MHC molecules could bind and present
more self-antigens. This would require the elimination of many
more self-reactive T cells during development (Chapter 20). Thus
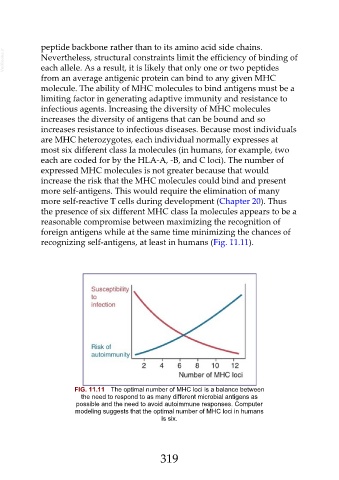
the presence of six different MHC class Ia molecules appears to be a
reasonable compromise between maximizing the recognition of
foreign antigens while at the same time minimizing the chances of
recognizing self-antigens, at least in humans (Fig. 11.11).
FIG. 11.11 The optimal number of MHC loci is a balance between
the need to respond to as many different microbial antigens as
possible and the need to avoid autoimmune responses. Computer
modeling suggests that the optimal number of MHC loci in humans
is six.
319