Page 45 - The Toxicology of Fishes
P. 45
Bioavailability of Chemical Contaminants in Aquatic Systems 25
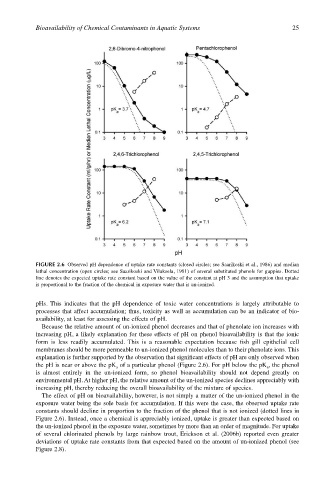
FIGURE 2.6 Observed pH dependence of uptake rate constants (closed circles; see Saarikoski et al., 1986) and median
lethal concentration (open circles; see Saarikoski and Viluksela, 1981) of several substituted phenols for guppies. Dotted
line denotes the expected uptake rate constant based on the value of the constant at pH 3 and the assumption that uptake
is proportional to the fraction of the chemical in exposure water that is un-ionized.
pHs. This indicates that the pH dependence of toxic water concentrations is largely attributable to
processes that affect accumulation; thus, toxicity as well as accumulation can be an indicator of bio-
availability, at least for assessing the effects of pH.
Because the relative amount of un-ionized phenol decreases and that of phenolate ion increases with
increasing pH, a likely explanation for these effects of pH on phenol bioavailability is that the ionic
form is less readily accumulated. This is a reasonable expectation because fish gill epithelial cell
membranes should be more permeable to un-ionized phenol molecules than to their phenolate ions. This
explanation is further supported by the observation that significant effects of pH are only observed when
the pH is near or above the pK of a particular phenol (Figure 2.6). For pH below the pK , the phenol
a
a
is almost entirely in the un-ionized form, so phenol bioavailability should not depend greatly on
environmental pH. At higher pH, the relative amount of the un-ionized species declines appreciably with
increasing pH, thereby reducing the overall bioavailability of the mixture of species.
The effect of pH on bioavailability, however, is not simply a matter of the un-ionized phenol in the
exposure water being the sole basis for accumulation. If this were the case, the observed uptake rate
constants should decline in proportion to the fraction of the phenol that is not ionized (dotted lines in
Figure 2.6). Instead, once a chemical is appreciably ionized, uptake is greater than expected based on
the un-ionized phenol in the exposure water, sometimes by more than an order of magnitude. For uptake
of several chlorinated phenols by large rainbow trout, Erickson et al. (2006b) reported even greater
deviations of uptake rate constants from that expected based on the amount of un-ionized phenol (see
Figure 2.8).