Page 46 - The Toxicology of Fishes
P. 46
26 The Toxicology of Fishes
500 1500 200
96-Hour Median Lethal Concentration (µg PCP/L) 300 32-Day Bioconcentration Factor (mL/g) 700 100 Estimated Lethal Body Burden (µg/g)
1000
150
200
500
150
100
70 300 70
200 50
7 8
pH
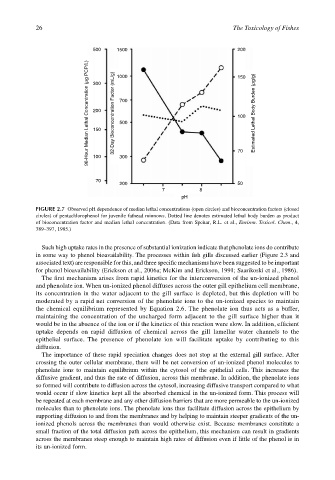
FIGURE 2.7 Observed pH dependence of median lethal concentrations (open circles) and bioconcentration factors (closed
circles) of pentachlorophenol for juvenile fathead minnows. Dotted line denotes estimated lethal body burden as product
of bioconcentration factor and median lethal concentration. (Data from Spehar, R.L. et al., Environ. Toxicol. Chem., 4,
389–397, 1985.)
Such high uptake rates in the presence of substantial ionization indicate that phenolate ions do contribute
in some way to phenol bioavailability. The processes within fish gills discussed earlier (Figure 2.3 and
associated text) are responsible for this, and three specific mechanisms have been suggested to be important
for phenol bioavailability (Erickson et al., 2006a; McKim and Erickson, 1991; Saarikoski et al., 1986).
The first mechanism arises from rapid kinetics for the interconversion of the un-ionized phenol
and phenolate ion. When un-ionized phenol diffuses across the outer gill epithelium cell membrane,
its concentration in the water adjacent to the gill surface is depleted, but this depletion will be
moderated by a rapid net conversion of the phenolate ions to the un-ionized species to maintain
the chemical equilibrium represented by Equation 2.6. The phenolate ion thus acts as a buffer,
maintaining the concentration of the uncharged form adjacent to the gill surface higher than it
would be in the absence of the ion or if the kinetics of this reaction were slow. In addition, efficient
uptake depends on rapid diffusion of chemical across the gill lamellar water channels to the
epithelial surface. The presence of phenolate ion will facilitate uptake by contributing to this
diffusion.
The importance of these rapid speciation changes does not stop at the external gill surface. After
crossing the outer cellular membrane, there will be net conversion of un-ionized phenol molecules to
phenolate ions to maintain equilibrium within the cytosol of the epithelial cells. This increases the
diffusive gradient, and thus the rate of diffusion, across this membrane. In addition, the phenolate ions
so formed will contribute to diffusion across the cytosol, increasing diffusive transport compared to what
would occur if slow kinetics kept all the absorbed chemical in the un-ionized form. This process will
be repeated at each membrane and any other diffusion barriers that are more permeable to the un-ionized
molecules than to phenolate ions. The phenolate ions thus facilitate diffusion across the epithelium by
supporting diffusion to and from the membranes and by helping to maintain steeper gradients of the un-
ionized phenols across the membranes than would otherwise exist. Because membranes constitute a
small fraction of the total diffusion path across the epithelium, this mechanism can result in gradients
across the membranes steep enough to maintain high rates of diffusion even if little of the phenol is in
its un-ionized form.