Page 47 - The Toxicology of Fishes
P. 47
Bioavailability of Chemical Contaminants in Aquatic Systems 27
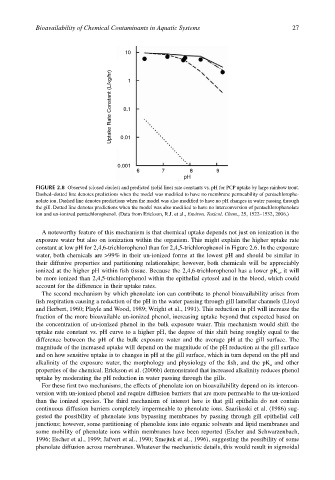
10
Uptake Rate Constant (L/kg/hr) 0.1 1
0.01
0.001
6 7 8 9
pH
FIGURE 2.8 Observed (closed circles) and predicted (solid line) rate constants vs. pH for PCP uptake by large rainbow trout.
Dashed–dotted line denotes predictions when the model was modified to have no membrane permeability of pentachlorophe-
nolate ion. Dashed line denotes predictions when the model was also modified to have no pH changes in water passing through
the gill. Dotted line denotes predictions when the model was also modified to have no interconversion of pentachlorophenolate
ion and un-ionized pentachlorophenol. (Data from Erickson, R.J. et al., Environ. Toxicol. Chem., 25, 1522–1532, 2006.)
A noteworthy feature of this mechanism is that chemical uptake depends not just on ionization in the
exposure water but also on ionization within the organism. This might explain the higher uptake rate
constant at low pH for 2,4,6-trichlorophenol than for 2,4,5-trichlorophenol in Figure 2.6. In the exposure
water, both chemicals are >99% in their un-ionized forms at the lowest pH and should be similar in
their diffusive properties and partitioning relationships; however, both chemicals will be appreciably
ionized at the higher pH within fish tissue. Because the 2,4,6-trichlorophenol has a lower pK , it will
a
be more ionized than 2,4,5-trichlorophenol within the epithelial cytosol and in the blood, which could
account for the difference in their uptake rates.
The second mechanism by which phenolate ion can contribute to phenol bioavailability arises from
fish respiration causing a reduction of the pH in the water passing through gill lamellar channels (Lloyd
and Herbert, 1960; Playle and Wood, 1989; Wright et al., 1991). This reduction in pH will increase the
fraction of the more bioavailable un-ionized phenol, increasing uptake beyond that expected based on
the concentration of un-ionized phenol in the bulk exposure water. This mechanism would shift the
uptake rate constant vs. pH curve to a higher pH, the degree of this shift being roughly equal to the
difference between the pH of the bulk exposure water and the average pH at the gill surface. The
magnitude of the increased uptake will depend on the magnitude of the pH reduction at the gill surface
and on how sensitive uptake is to changes in pH at the gill surface, which in turn depend on the pH and
alkalinity of the exposure water, the morphology and physiology of the fish, and the pK and other
a
properties of the chemical. Erickson et al. (2006b) demonstrated that increased alkalinity reduces phenol
uptake by moderating the pH reduction in water passing through the gills.
For these first two mechanisms, the effects of phenolate ion on bioavailability depend on its intercon-
version with un-ionized phenol and require diffusion barriers that are more permeable to the un-ionized
than the ionized species. The third mechanism of interest here is that gill epithelia do not contain
continuous diffusion barriers completely impermeable to phenolate ions. Saarikoski et al. (1986) sug-
gested the possibility of phenolate ions bypassing membranes by passing through gill epithelial cell
junctions; however, some partitioning of phenolate ions into organic solvents and lipid membranes and
some mobility of phenolate ions within membranes have been reported (Escher and Schwarzenbach,
1996; Escher et al., 1999; Jafvert et al., 1990; Smejtek et al., 1996), suggesting the possibility of some
phenolate diffusion across membranes. Whatever the mechanistic details, this would result in sigmoidal