Page 19 - EXOSOMES
P. 19
www.advancedsciencenews.com www.small-journal.com
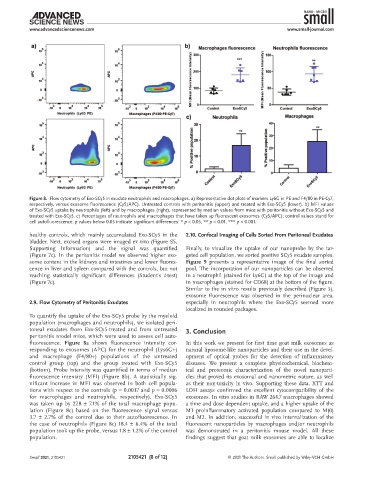
Figure 8. Flow cytometry of Exo-SCy5 in exudate neutrophils and macrophages. a) Representative dot plots of markers Ly6G in PE and F4/80 in PE-Cy7,
respectively, versus exosome fluorescence (Cy5/APC). Untreated controls with peritonitis (upper) and treated with Exo-SCy5 (lower). b) MFI values
of Exo-SCy5 uptake by neutrophils (left) and by macrophages (right), represented by median values from mice with peritonitis without Exo-SCy5 and
treated with Exo-SCy5. c) Percentages of neutrophils and macrophages that have taken up fluorescent exosomes (Cy5/APC); control values stand for
cell autofluorescence. p values below 0.05 indicate significant differences: * p < 0.05, ** p < 0.01, *** p < 0.001.
healthy controls, which mainly accumulated Exo-SCy5 in the 2.10. Confocal Imaging of Cells Sorted From Peritoneal Exudates
bladder. Next, excised organs were imaged ex vivo (Figure S5,
Supporting Information) and the signal was quantified Finally, to visualize the uptake of our nanoprobe by the tar-
(Figure 7c). In the peritonitis model we observed higher exo- geted cell population, we sorted positive SCy5 exudate samples.
some content in the kidneys and intestines and lower fluores- Figure 9 presents a representative image of the final sorted
cence in liver and spleen compared with the controls, but not pool. The incorporation of our nanoparticles can be observed
reaching statistically significant differences (Student’s t-test) in a neutrophil (stained for Ly6G) at the top of the image and
(Figure 7c). in macrophages (stained for CD68) at the bottom of the figure.
Similar to the in vitro results previously described (Figure 5),
exosome fluorescence was observed in the perinuclear area,
2.9. Flow Cytometry of Peritonitis Exudates especially in neutrophils where the Exo-SCy5 seemed more
localized in rounded packages.
To quantify the uptake of the Exo-SCy5 probe by the myeloid
population (macrophages and neutrophils), we isolated peri-
toneal exudates from Exo-SCy5-treated and from untreated 3. Conclusion
peritonitis model mice, which were used to assess cell auto-
fluorescence. Figure 8a shows fluorescence intensity cor- In this work we present for first time goat milk exosomes as
responding to exosomes (APC) for the neutrophil (Lys6G+) natural liposome-like nanoparticles and their use in the devel-
and macrophage (F4/80+) populations of the untreated opment of optical probes for the detection of inflammatory
control group (top) and the group treated with Exo-SCy5 diseases. We present a complete physicochemical, biochem-
(bottom). Probe intensity was quantified in terms of median ical and proteomic characterization of the novel nanoparti-
fluorescence intensity (MFI) (Figure 8b). A statistically sig- cles that proved its exosomal and nanometric nature, as well
nificant increase in MFI was observed in both cell popula- as their non-toxicity in vivo. Supporting these data, XTT and
tions with respect to the controls (p = 0.0037 and p = 0.0006 LDH assays confirmed the excellent cytocompatibility of the
for macrophages and neutrophils, respectively). Exo-SCy5 exosomes. In vitro studies in RAW 264.7 macrophages showed
was taken up by 27.8 ± 7.1% of the total macrophage popu- a time and dose dependent uptake, and a higher uptake of the
lation (Figure 8c) based on the fluorescence signal versus M1 proinflammatory activated population compared to M(0)
3.7 ± 2.7% of the control due to their autofluorescence. In and M2. In addition, successful in vivo internalization of the
the case of neutrophils (Figure 8c) 18.4 ± 6.4% of the total fluorescent nanoparticles by macrophages and/or neutrophils
population took up the probe, versus 1.8 ± 1.2% of the control was demonstrated in a peritonitis mouse model. All these
population. findings suggest that goat milk exosomes are able to localize
Small 2021, 2105421 2105421 (8 of 12) © 2021 The Authors. Small published by Wiley-VCH GmbH