Page 31 - Instrumental Analysis - Pharm D Clinical- 07-PA403
P. 31
UV-visible Spectrophotometry
3- Chemical deviations
• Type of solvent: Most compounds show different absorption spectra and
λmax values in different solvents. All standard and sample solutions must be
prepared in the same solvent otherwise erroneous results will be obtained.
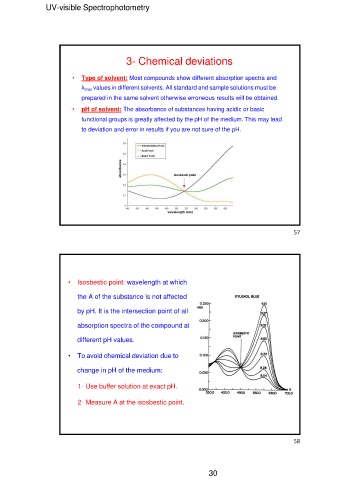
• pH of solvent: The absorbance of substances having acidic or basic
functional groups is greatly affected by the pH of the medium. This may lead
to deviation and error in results if you are not sure of the pH.
• Isosbestic point: wavelength at which 57
the A of the substance is not affected 58
by pH. It is the intersection point of all
absorption spectra of the compound at
different pH values.
• To avoid chemical deviation due to
change in pH of the medium:
1- Use buffer solution at exact pH.
2- Measure A at the isosbestic point.
30