Page 35 - Instrumental Analysis - Pharm D Clinical- 07-PA403
P. 35
UV-visible Spectrophotometry
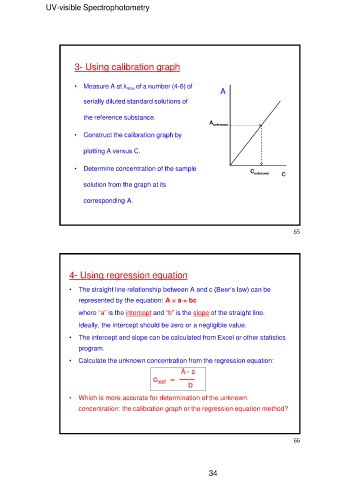
3- Using calibration graph
• Measure A at λmax of a number (4-6) of A
serially diluted standard solutions of
the reference substance. Aunknown
• Construct the calibration graph by
plotting A versus C.
• Determine concentration of the sample Cunknown c
solution from the graph at its
corresponding A.
65
4- Using regression equation
• The straight line relationship between A and c (Beer’s law) can be
represented by the equation: A = a + bc
where “a” is the intercept and “b” is the slope of the straight line.
Ideally, the intercept should be zero or a negligible value.
• The intercept and slope can be calculated from Excel or other statistics
program.
• Calculate the unknown concentration from the regression equation:
A‒ a
────
ctest = b
• Which is more accurate for determination of the unknown
concentration: the calibration graph or the regression equation method?
66
34