Page 40 - Instrumental Analysis - Pharm D Clinical- 07-PA403
P. 40
Spectrofluorimetry
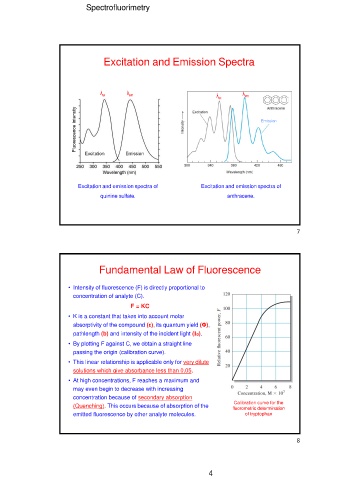
Excitation and Emission Spectra
λex λem λex λem
Fluorescence intensity Emission
Excitation Emission
Excitation and emission spectra of Excitation and emission spectra of
quinine sulfate. anthracene.
7
Fundamental Law of Fluorescence
• Intensity of fluorescence (F) is directly proportional to Calibration curve for the
concentration of analyte (C). fluorometric determination
F = KC of tryptophan
• K is a constant that takes into account molar
absorptivity of the compound (ε), its quantum yield (Φ),
pathlength (b) and intensity of the incident light (I0).
• By plotting F against C, we obtain a straight line
passing the origin (calibration curve).
• This linear relationship is applicable only for very dilute
solutions which give absorbance less than 0.05.
• At high concentrations, F reaches a maximum and
may even begin to decrease with increasing
concentration because of secondary absorption
(Quenching). This occurs because of absorption of the
emitted fluorescence by other analyte molecules.
8
4