Page 852 - Veterinary Toxicology, Basic and Clinical Principles, 3rd Edition
P. 852
810 SECTION | XIII Estrogenic Toxicants
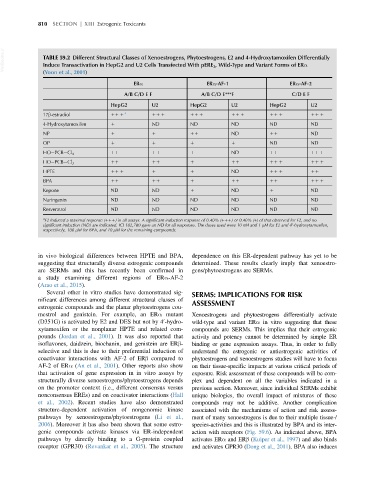
VetBooks.ir TABLE 59.2 Different Structural Classes of Xenoestrogens, Phytoestrogens, E2 and 4-Hydroxytamoxifen Differentially
Induce Transactivation in HepG2 and U2 Cells Transfected With pERE 3 , Wild-Type and Variant Forms of ERα
(Yoon et al., 2001)
ERα ERα-AF-1 ERα-AF-2
A/B C/D E F A/B C/D E***F C/D E F
HepG2 U2 HepG2 U2 HepG2 U2
17β-estradiol 11 1 a 11 1 11 1 11 1 11 1 11 1
4-Hydroxytamoxifen 1 ND ND ND ND ND
NP 1 1 11 ND 11 ND
OP 1 1 1 1 ND ND
11 11 1 ND 11 11 1
HO PCB Cl 4
HO PCB Cl 3 11 11 1 11 11 1 11 1
HPTE 11 1 1 1 ND 11 1 11
BPA 11 11 1 11 11 11 1
Kepone ND ND 1 ND 1 ND
Naringenin ND ND ND ND ND ND
Resveratrol ND ND ND ND ND ND
a
E2 induced a maximal response (111) in all assays. A significant induction response of 0.40% (111) or 0.40% (1) of that observed for E2, and no
significant induction (ND) are indicated. ICI 182,780 gave an ND for all responses. The doses used were 10 nM and 1 μM for E2 and 4 -hydroxytamoxifen,
0
respectively, 100 μM for BPA, and 10 μM for the remaining compounds.
in vivo biological differences between HPTE and BPA, dependence on this ER-dependent pathway has yet to be
suggesting that structurally diverse estrogenic compounds determined. These results clearly imply that xenoestro-
are SERMs and this has recently been confirmed in gens/phytoestrogens are SERMs.
a study examining different regions of ERα-AF-2
(Arao et al., 2015).
Several other in vitro studies have demonstrated sig- SERMS: IMPLICATIONS FOR RISK
nificant differences among different structural classes of ASSESSMENT
estrogenic compounds and the planar phytoestrogens cou-
mestrol and genistein. For example, an ERα mutant Xenoestrogens and phytoestrogens differentially activate
(D351G) is activated by E2 and DES but not by 4 -hydro- wild-type and variant ERα in vitro suggesting that these
0
xytamoxifen or the nonplanar HPTE and related com- compounds are SERMs. This implies that their estrogenic
pounds (Jordan et al., 2001). It was also reported that activity and potency cannot be determined by simple ER
isoflavones, daidzein, biochanin, and genistein are ERβ- binding or gene expression assays. Thus, in order to fully
selective and this is due to their preferential induction of understand the estrogenic or antiestrogenic activities of
coactivator interactions with AF-2 of ERβ compared to phytoestrogens and xenoestrogens studies will have to focus
AF-2 of ERα (An et al., 2001). Other reports also show on their tissue-specific impacts at various critical periods of
that activation of gene expression in in vitro assays by exposure. Risk assessment of these compounds will be com-
structurally diverse xenoestrogens/phytoestrogens depends plex and dependent on all the variables indicated in a
on the promoter context (i.e., different consensus versus previous section. Moreover, since individual SERMs exhibit
nonconsensus EREs) and on coactivator interactions (Hall unique biologies, the overall impact of mixtures of these
et al., 2002). Recent studies have also demonstrated compounds may not be additive. Another complication
structure-dependent activation of nongenomic kinase associated with the mechanisms of action and risk assess-
pathways by xenoestrogens/phytoestrogens (Li et al., ment of many xenoestrogens is due to their multiple tissue-/
2006). Moreover it has also been shown that some estro- species-activities and this is illustrated by BPA and its inter-
genic compounds activate kinases via ER-independent action with receptors (Fig. 59.6). As indicated above, BPA
pathways by directly binding to a G-protein coupled activates ERα and ERβ (Kuiper et al., 1997) and also binds
receptor (GPR30) (Revankar et al., 2005). The structure and activates GPR30 (Dong et al., 2011). BPA also induces