Page 534 - Veterinary Toxicology, Basic and Clinical Principles, 3rd Edition
P. 534
Organophosphates and Carbamates Chapter | 37 501
VetBooks.ir (H C) HCO O F HC O O F
)
C(CH 3 3
P
P
3
2
CH 3 CH 3
CH 3
Soman (GD)
Sarin (GB)
Isopropyl methylphosphonofluoridate Pinacolyl methylphosphonofluoridate
H C
3
O
O CH CH 2
H C P S CH 2 CH 2 N
H O CN 3
C 2 5 P
CH CH 3
) OCH 2 CH 3
N(CH 3 2
H 3 C
Tabun (GA)
Ethyl N-dimethyl phosphoramido-cyanidate O-ethyl-S-(2-diisopropylaminoethyl)
methylphosphonothiolate (VX)
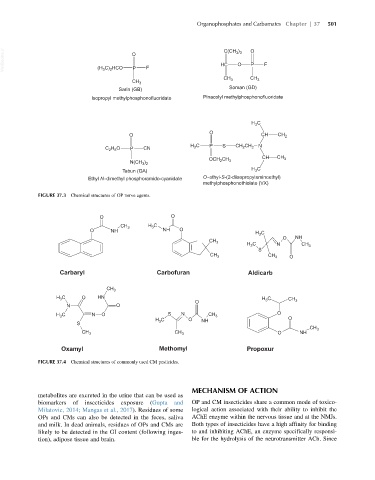
FIGURE 37.3 Chemical structures of OP nerve agents.
O O
H C
CH 3 3
O NH NH O
H C
3
O NH
CH 3
H 3 C N CH 3
S
CH 3 CH 3 O
Carbaryl Carbofuran Aldicarb
CH 3
H 3 C O HN H 3 C CH
O 3
N O
H 3 C N O S N CH 3 O
H C O NH O
S 3
CH 3
CH 3 CH 3 O NH
Oxamyl Methomyl Propoxur
FIGURE 37.4 Chemical structures of commonly used CM pesticides.
MECHANISM OF ACTION
metabolites are excreted in the urine that can be used as
biomarkers of insecticides exposure (Gupta and OP and CM insecticides share a common mode of toxico-
Milatovic, 2014; Mangas et al., 2017). Residues of some logical action associated with their ability to inhibit the
OPs and CMs can also be detected in the feces, saliva AChE enzyme within the nervous tissue and at the NMJs.
and milk. In dead animals, residues of OPs and CMs are Both types of insecticides have a high affinity for binding
likely to be detected in the GI content (following inges- to and inhibiting AChE, an enzyme specifically responsi-
tion), adipose tissue and brain. ble for the hydrolysis of the neurotransmitter ACh. Since