Page 49 - Essential Haematology
P. 49
Chapter 3 Hypochromic anaemias / 35
Daily loss ~ 1mg
Urine, faeces,
nails, hair, skin
Duodenum
Daily absorption Liver, other parenchymal
~ 1 mg cells and tissues,
especially muscle myoglobin
(600–650 mg)
Transferrin
Plasma
(4 mg)
Bone marrow
normoblasts
(150 mg)
Ineffective erythropoiesis
Macrophages
(0.5–1.5 g)
Circulating
haemoglobin
(1.7–2.4 g)
Menstrual loss
(haemorrhage)
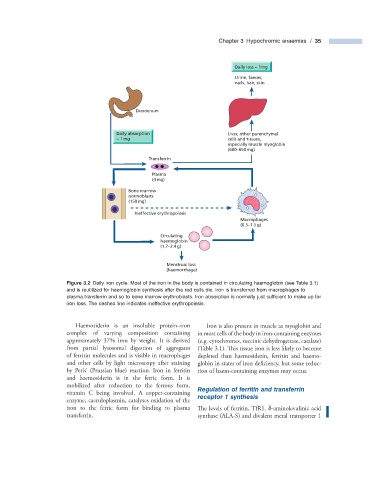
Figure 3.2 Daily iron cycle. Most of the iron in the body is contained in circulating haemoglobin (see Table 3.1 )
and is reutilized for haemoglobin synthesis after the red cells die. Iron is transferred from macrophages to
plasma transferrin and so to bone marrow erythroblasts. Iron absorption is normally just suffi cient to make up for
iron loss. The dashed line indicates ineffective erythropoiesis.
Haemosiderin is an insoluble protein – iron Iron is also present in muscle as myoglobin and
complex of varying composition containing in most cells of the body in iron - containing enzymes
approximately 37% iron by weight. It is derived (e.g. cytochromes, succinic dehydrogenase, catalase)
from partial lysosomal digestion of aggregates (Table 3.1 ). This tissue iron is less likely to become
of ferritin molecules and is visible in macrophages depleted than haemosiderin, ferritin and haemo-
and other cells by light microscopy after staining globin in states of iron deficiency, but some reduc-
by Perls ’ (Prussian blue) reaction. Iron in ferritin tion of haem - containing enzymes may occur.
and haemosiderin is in the ferric form. It is
mobilized after reduction to the ferrous form,
vitamin C being involved. A copper - containing Regulation of f erritin and t ransferrin
enzyme, caeruloplasmin, catalyses oxidation of the r eceptor 1 s ynthesis
iron to the ferric form for binding to plasma The levels of ferritin, TfR1, δ - aminolevulinic acid
transferrin. synthase (ALA - S) and divalent metal transporter 1